|
Through a collaboration with research groups in Biomedical Engineering and the Medical Sciences,
we are performing atomic-force microscopy studies of biological cells in order to better
understand their structure, mechanical properties, and adhesive properties. For instance, we are
measuring the adhesion of cells at the single-bond level, and have developed techniques to
characterize their response to chemical stimulants.
|
|
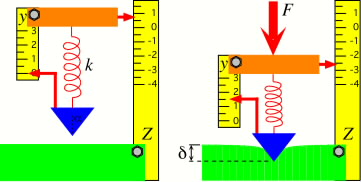
AFM model showing cantilever deflection (modeled here as the compression of a
spring) resulting from indentation of a soft surface. The sample deformation is inferred from the
meaured deflection and known sample displacement.
|
| | | |
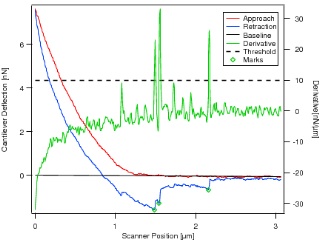
Typical force curve seen for a fibronecting-coated bead making contact with
a live NIH-3T3 mouse fibroblast. Several bond-rupture events are seen, marked by green circles.
|
|
The adhesion of biological cells to surfaces is a complex process that is critically important to
the cell's ability to migrate and to form tissue. We are using the AFM to study the biology and
physics of cellular adhesion to extra-cellular matrix proteins such as fibronectin, and the effects
of stimulants on this interaction. A better understanding of cellular adhesive properties and
motion will aid in the design of active wound-dressing materials as well as improve our basic
understanding of cellular properties.
|
| | | |
|
The AFM can also be used to study the mechanical properties of a sample surface by measuring the
deformation due to an applied force. The extreme sensitivity of the AFM to small forces permits
adhesion and mechanical properties to be measured for materials as soft as live cells and as small
as carbon nanotubes.
|
|

Height, deflection, and elastic modulus maps of (a) human rhabdomyosarcoma cells
and (b) NIH-3T3 mouse fibroblasts. The scale bar measures 20 µm.
|


